|
The state of the atom, in this case, is called the Ground-state where the radius of the orbit (a 0) is the least. The energy levels gradually increase in the outer orbits, when n = 2, 3, … 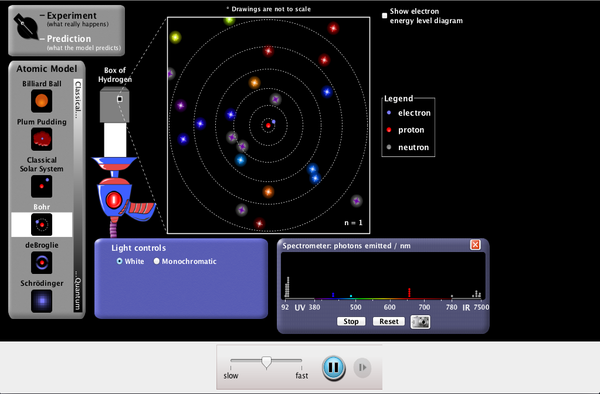
The energy of an atom is the lowest when its electron is revolving in an orbit closest to the nucleus (i.e. The negative sign denotes that the electron is bound to the nucleus. The total energy of an electron in Bohr’s stationary orbit The velocity of the electron in Bohr’s stationary orbit Z is the atomic number of the atom (Z = 1 for hydrogen atom), It violates the Heisenberg Uncertainty Principle.The spectra obtained from larger atoms weren’t explained.It failed to explain the Stark effect (effect of electric field on the spectra of atoms).Bohr’s model of the atom failed to explain the Zeeman Effect (effect of magnetic field on the spectra of atoms).(Etis the initial state and Efis the final state and Et>Ef) The frequency of the released photon is presented as, Then, a photon is released having energy equal to energy difference between the initial and final states. Postulate III: An electron might make transitions from one of its specified non-radiating orbits to another of lower energy. 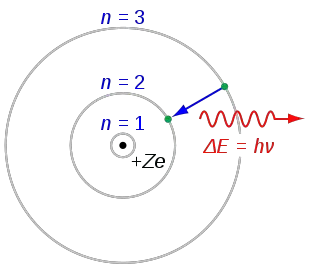
The angular momentum of orbiting electron is calculated by L=nh/2 Postulate II: The electron revolves around the nucleus only in those orbits for which the angular momentum is some integral multiple of h/2. These are called the stationary states of atoms Postulate I: An electron in an atom could revolve in certain stable orbits without emission of radiant energy.Īccording to Bohr, each atom can exist in certain definite stable states and each state has fixed total energy. Thus, a continuous spectrum can be observed.īohr defined his model using three postulates applying the concepts of classical electromagnetic theory and initial studies on quantum theory together. The angular velocity and frequency of such electrons will keep on changing, alternating the frequency of light released. The theory also states that the frequency of the electromagnetic waves discharged by the revolving electrons will be equal to that of the revolution. An atom, in such a case, cannot be stable. The electron will then revolve towards the nucleus. The energy of an accelerating electron must be decreasing. In addition, it is known that there is a constant acceleration of an object moving in a circle due to centripetal force.įurthermore, the electromagnetic theory says that an accelerating charged particle will emit radiation in the form of electromagnetic waves. This is because both the nucleus and electrons are charged particles. 
But, the elementary difference between the two situations is that the planetary system is held by the gravitational force whereas the nucleus-electron system interacts by the Coulomb’s Law of Force. Rutherford had assumed the atom and its components work in a manner similar to the solar system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
